Protein Aggregation and Autophagy
Centre de génétique et de Physiologie Moélculaires et cellulairesCNRS UMR5534
16 Rue Dubois
Campus de la Doua
69622 Villeurbanne Cedex - Villeurbanne
Site web - carole.kretz@univ-lyon1.fr -
Principal investigator
Pr Carole Kretz-Remy

Research themes
Whether a protein folds correctly or whether it aggregates is dependent on mutations or modifications but can also be promoted by pH modulation, UV irradiation or heat stresses. Since aggregates can trap transcription factors and/or cytosolic signaling components, they are often toxic for the cell. Cells have therefore adapted various quality control mechanisms to minimize misfolding and to eliminate misfolded and/or aggregated proteins. The three major mechanisms of protein quality control are: molecular chaperones, the ubiquitin-proteasome system and autophagy/lysosome process.
Our group is interested in studying cell response to protein aggregation stresses. We demonstrated that protein aggregation activates NFκB transcription factor that, in turn, activates autophagy. This latter mechanism helps the cells to get rid off protein aggregates. We are studying two aspects of this response.
- NFκB and autophagy involvement in protein quality control. We are determining the molecular mechanisms by which protein aggregates activate NFκB-dependent autophagy in order to identify actors that can modulate the NFκB-dependent autophagy pathway, for therapeutic prospects (protein conformational diseases, anti-cancer treatment by hyperthermia).
- Muscle degeneration and protein quality control. Myopathies are often associated with protein aggregation and/or mitochondrial disorders and modifications of the autophagic process. We aim at characterizing quality control in cellular models of Duchenne’s Muscular Dystrophy, focusing on NFκB, protein aggregates, autophagy and mitochondria. This work is performed in collaboration with Professor Kathrin Gieseler’s Lab (Dr Ludivine Walter).
Our work is supported by the « Université Claude Bernard Lyon I », the « CNRS », the « Comité départemental du Rhône de la Ligue contre le Cancer » and the « Fondation pour la Recherche Médicale ».
Descriptive figure
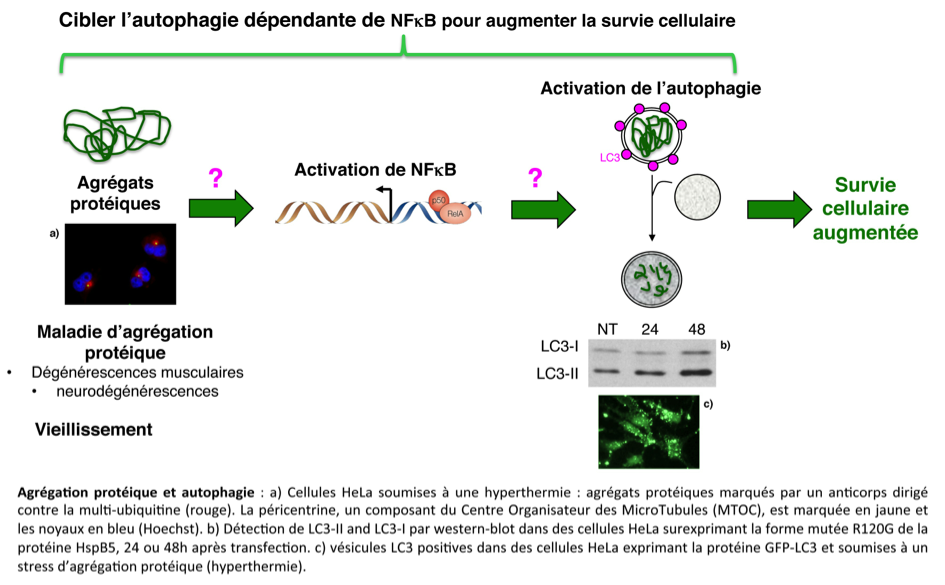
Publications
• Nivon M., Abou-Samra M., Richet E., Arrigo A.-P. and C Kretz-Remy (2012) NF-κB regulates protein quality control after heat stress though modulation of Bag3/HspB8 complex. J Cell Sci, 125, 1141-51.
• Nivon, M., Richet, E., Codogno, P. and C. Kretz-Remy (2009) Autophagy activation by NF-κB is essential for cell survival after heat shock. Autophagy, 5, 766-83.
• Firdaus, W.J.J., Wyttenbach A., Giuliano, P., Kretz-Remy, C., Currie, R.W. and A.-P. Arrigo (2006) Huntingtin inclusion bodies are iron-dependent centers of oxidative events. FEBS J, 273, 5428-41.
• Kretz-Remy C., Munsch B., and A.-P. Arrigo (2001) κB-dependent transcriptional activation during heat shock recovery : thermolability of the NF-κB/IκB complex. J. Biol. Chem., 276,43723-33.
Composition de l'équipe
Pascale Muller, adjointe technique UCB Lyon I
Mathieu Baritaud, post-doctorant (FRM)
Marion Wattin, étudiante M2R (UCB Lyon I)