Pathophysiology of parkinsonian syndromes
Institut des Maladies NeurodégénérativesUniversité de Bordeaux
CNRS UMR 5293 – Bât. Neurocampus
146 Rue Léo Saignat
33076 Bordeaux – France - Bordeaux
Site web - benjamin.dehay@u-bordeaux.fr -
Principal investigator
Benjamin Dehay

Research themes
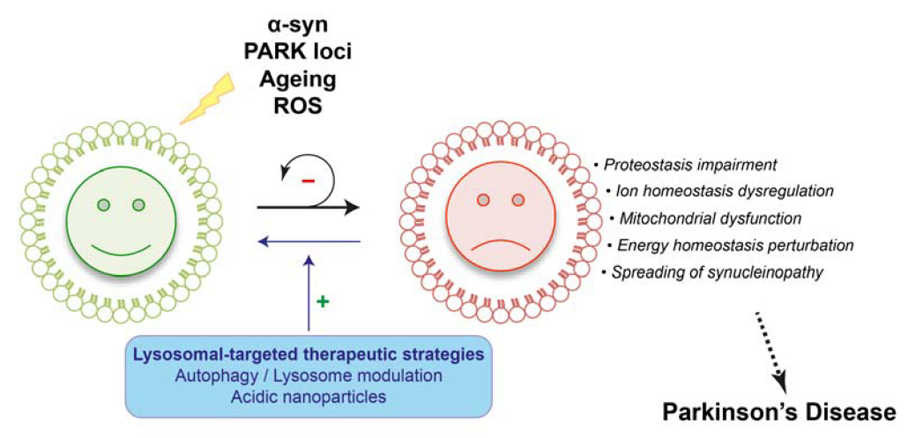
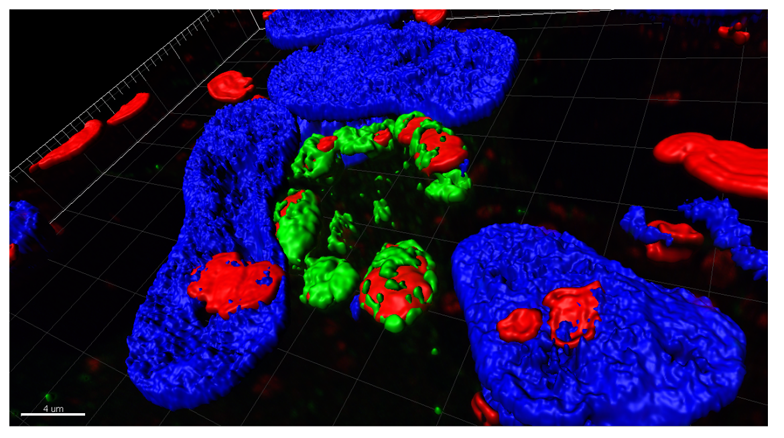
Parkinson’s disease is characterized by a progressive accumulation and propagation of a-synuclein likely responsible for their subsequent death. Our objective is to understand these mechanisms for then proposing adapted therapeutic approaches. Increasing evidence indicates that impairment of autophagy-lysosomal function may contribute to the pathogenesis of Parkinson’s disease. In particular, lysosomal defects could potentially account not only for neuronal cell dysfunction/death, the presence of α-synuclein-containing Lewy bodies, but also for the α-synuclein release from neurons. Our main goal is to understand how the disease proteins cause alterations of the proteolytic pathways contributing to the pathogenesis and, more importantly, whether functional augmentation of the lysosomal pathway can lead to clearance of the toxic proteins and halt the disease process. For this second purpose, we develop new neuroprotective/disease-modifying therapeutic strategies aimed at restoring lysosomal levels and function towards multidimensional clinically-relevant rodent and primate models. In addition to the various research projects, we begin the study of the interaction between autophagy and ciliary machinery in the context of neurodegenerative disease, with a special focus on Alzheimer’s disease.
Descriptive figure
Publications
Bourdenx M, Dehay B. Autophagy and brain: specificities and dysfunctions. Med Sci (Paris), volume 33 n°3 (2017).
Bourdenx M, Dehay B. What lysosomes actually tell us about Parkinson’s disease? Ageing Res Rev. 2016 Mar3. pii:S1568-1637(16)30023-X.
Bourdenx M, Daniel J, Genin E, Soria FN, Blanchard-Desce M, Bezard E, Dehay B#. Nanoparticles restore lysosomal acidification defects: Implication for Parkinson’s and other lysosomal-related diseases. Autophagy. 2016 Mar 3;12(3):472-83.
Bourdenx, M., Bezard, E., Dehay, B. Lysosomes and α-synuclein form a dangerous duet leading to neuronal cell death. Front Neuroanat. 2014 Aug 14;8:83.
Bove, J. †, Martinez-Vicente, M. †, Dehay, B. †, Perier, C., Recasens, A., Bombrun, A., Antonsson, B., and Vila, M. Bax channel activity mediates lysosomal disruption linked to Parkinson’s disease. Autophagy. 2014 May;10(5):889-900. († co-first author)
Dehay, B., Martinez-Vicente, M., Caldwell, G., Caldwell, K., Yue, Z., Cookson, M., Klein, C., Vila, M., and Bezard, E. Lysosomal Impairment in Parkinson’s disease. Mov Disorder. 2013 Jun;28(6):725-32.
Dehay, B., Martinez-Vicente, M., Ramirez, A., Perier, C., Klein, C., Vila, M., and Bezard, E. Lysosomal dysfunction in Parkinson disease: ATP13A2 gets into the groove. Autophagy. 2012. Sep;8(9):1389-91.
Dehay, B., Ramirez, A., Martinez-Vicente, M., Perier, C., Canron, MH., Doudnikoff, E., Vital, A., Vila, M., Klein, C., and Bezard, E. Loss of P-type ATPase ATP13A2/PARK9 function induces general lysosomal deficiency and leads to Parkinson disease neurodegeneration. Proc Natl Acad Sci U S A. 2012 Jun 12;109 (24) :9611-6.
Vila, M., Bove, J., Dehay, B., Rodriguez-Muela, C., and Boya, P. Lysosomal membrane permeabilization in Parkinson’s Disease. Autophagy 2011 Jan;7(1):98-100.
Dehay, B., Bove, J., Rodriguez-Muela, C., Perier, C., Recasens, A., Boya, P., and Vila, M. Pathogenic lysosomal depletion in Parkinson’s Disease. J Neurosci. 2010 Sep 15; 30(37): 12535-44.
Composition de l'équipe
AROTCARENA Marie-Laure (doctorante)
BEZARD Erwan (chercheur)
BOUE-GRABOT Eric (chercheur)
BOURDENX Mathieu (post-doctorant)
CANRON Marie-Hélène (assistante ingénieure)
CHANSEL Lucie (doctorante assistante HU)
DEHAY Benjamin (chercheur)
DOVERO Sandra (ingénieure d’étude)
DOUDNIKOFF Marie-Evelyne (assistante ingénieure)
ESTAGER Alain (adjoint technique)
FERNAGUT Pierre-Olivier (chercheur)
GUERIN Paul-Arnaud (doctorant)
JIMENEZ Clément (PU-PH)
LARGITTE Leslie-Ann (ingénieure d’étude)
MARTIN-NEGRIER Marie-Laure (PU-PH)
MARTINEZ Audrey (assistante ingénieure)
MASSE Karine (MCU)
MEISSNER Wassilios (PU-PH)
PAMPLIEGA Olatz (post-doctorante)
SORIA Federico (post-doctorant)
THIOLAT Marie-Laure (technicienne)
TISON François (PU-PH)
VITAL Anne (PU-PH)